
The origin of life has been one of the most fundamental questions of human existence, ever since our first self-conscious ancestors began to walk upright and gazed at the night sky. For millenia, the heavens were seen as being completely separate from the material world, forever out of reach to human experience. The advent of modern science helped to systematically erode this worldview by revealing a Universe where the same physical laws applied similarly on Earth as in the skies, while also establishing that both were made up from the exact same elements. In addition, the scientific study of the Universe has provided an ever-growing body of evidence during the last half century, which suggest that even the chemical building blocks of life itself were probably forged over many eons in the vast expanses between the stars before finally finding their way into the young Earth, shortly after its formation 4.5 billion years ago.
Now the latest observations from the Atacama Large Millimeter/submillimeter Array, or ALMA for short, in Chile, come to provide more credence to this hypothesis by revealing the presence of complex organic molecules inside the protoplanetary disk of a newly formed nearby star, which suggests that the building blocks for the development of life in the Universe could be readily available during the early stages of planetary formation and evolution.
Our current scientific understanding regarding the emergence of life on the primordial Earth posits that the latter was dependent on the presence of three main ingredients: water, energy, and organic (carbon-based) chemical compounds. In the search for life in the Universe it only makes sense for scientists to be looking for the same elements in other extraterrestrial environments. One of the great revelations of planetary exploration has been the realisation that all of the basic prerequisites for life are also abundant in many Solar System bodies besides Earth, like the ice moons of the outer gas giant planets, while minor bodies like comets and asteroids have been also found to be rich in organic material.
Furthermore, the advent of space-based infrared astronomy during the 1970s allowed scientists to peer through the veil of dust and gas of the interstellar medium (which is composed of all the matter that exists between the stars), opening a new window from which they could conduct detailed spectroscopic studies of astrophysical processes that were previously invisible. These, as well as additional studies of interstellar gas and dust clouds in other parts of the electromagnetic spectrum, have among other things helped to revel an ever-growing list of hundreds of organic molecules, many of which are surprisingly complex.
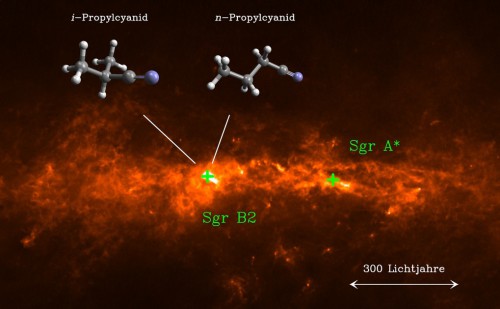
Several examples worthy of note are the discovery of glycine (which is the simplest of the 20 amino acids that are essential for human life) as well as cyanomethanimine, ethanamine, and iso-propyl cyanide (complex pre-biotic molecules that are part of our genetic structure) in Sagittarius B2, a massive molecular cloud located just 400 light-years from the center of the Milky Way. Life’s ingredients have also been found in various places far from the galactic center, with astronomers reporting in recent years the discoveries of formamide around the nearby protostar IRAS 16293-2422 and acetylene and hydrogen cyanide in the circumstellar disk around the young star IRS 46.
Now an international team of astronomers from the U.S., the Netherlands, and Japan, led by Karin Öberg from the Harvard-Smithsonian Center for Astrophysics in Cambridge, Mass., come to add further exciting evidence for the prevalence of the building blocks of life in the Universe, with the detection of methyl cyanide in the outskirts of the protoplanetary disk around MWC 480, a young, Sun-like star that has 10 times the luminosity and 1.8 times the mass of our home star. Located approximately 455 light-years away in the Taurus star forming region, one of the nearest such regions to our Solar System, MWC 480 is currently in the early stages of planetary formation, featuring a cicrcumstellar disk which is estimated to be about 1 million years old.

More specifically, Öberg’s team was able to detect two emission lines of methyl cyanide in MWC 480’s spectrum in addition to finding clear lines of cyanoacetylene as well. Both molecules are of huge biological importance to humans, since when combined with simpler compounds like water and ammonia they can lead to the formation of amino acids (which in turn are the building blocks of proteins) and nucleic acids like DNA and RNA which form the genetic code of all life on Earth. “Studies of comets and asteroids show that the solar nebula that spawned the Sun and planets was rich in water and complex organic compounds,” says Öberg. “We now have even better evidence that this same chemistry exists elsewhere in the Universe, in regions that could form solar systems not unlike our own.”
The superior observing capabilities of the ALMA array, which comprises a large interferometer of 66 high-precision antennas that act together as a single large radio dish, provided the researchers with the angular resolution needed in order to map the distribution of the organic compounds around MWC 480. The observations showed that the latter were spread over a wide area far from the star, between 30 and 100 Astronomical Units (approximately between 4.5 and 15 billion kilometers).
“We calculate abundances of methyl cyanide and cyanoacetylene with respect to hydrogen cyanide at 30 AU, the smallest disk radius accessible by the ALMA observations, and at 100 AU, the outer boundary of the cyanide emission maps,” write the researchers in their study which appeared in the journal Nature. “Accounting for the higher luminosity of MWC 480 compared to the young Sun, this radial range in the MWC 480 disk corresponds to the comet forming zone of [the Kuiper Belt] of 10–30 AU in the Solar Nebula.” More importantly, Öberg’s team noticed that the levels of methyl cyanide and cyanoacetylene were found to be higher than those observed comets in our own Solar System and were also much higher than those of the circumstellar disk around IRAS 16298-2422, a less evolved protostar which has an estimated age of 300,000 years.
These higher abundances indicate that more complex organic compounds can be produced inside protoplanetary disks in very short timescales of several hundred thousand years, before planets have time to fully form. The presence of these compounds in the outer regions of the protoplanetary disk around MWC 480 also suggests that the former could eventually be locked inside icy cometary bodies similar to the comets that are found in the Kuiper Belt of our own Solar System. These comets could in turn provide the basic ingredients of life to any life-friendly planets that may form in the star’s inner regions. “The high ratio of complex to simple cyanides in the MWC 480 disk implies that the rich organic composition of comets is not unique to our Solar System, and could be common-place,” conclude the researchers. “Laboratory experiments have shown that the same ice chemistry that produce methyl cyanide, i.e. photo processing of interstellar ice analogs also produce simple sugars and amino acids. This suggests that the early surface conditions of Earth, set by comet and asteroid bombardment, may be common for young rocky planets, and that conditions favourable to an even richer chemistry may be ubiquitous.”

These latest results from ALMA are in accordance with a previous observational study by a team of astronomers from the University of Hong Kong, which suggested that a series of aromatic and aliphatic hydrocarbons similar to coal and oil are widespread in the interstellar medium and can be produced from exploding stars within a timescale of a few weeks. Furthermore, these findings come in contrast to the predictions of established theoretical models which posit that such complex organic compounds cannot be formed in the vicinity of young and energetic stars due to the high levels of ionising stellar radiation which should destroy them in a short amount of time. “Studies of comets and asteroids show that the solar nebula that spawned our Sun and planets was rich in water and complex organic compounds,” says Öberg. “We now have evidence that this same chemistry exists elsewhere in the Universe, in regions that could form solar systems not unlike our own.”
The results of a recent independent study have reached a similar conclusion. Using the state-of-the-art Cosmic Simulation Chamber, or COSmIC for short, which is located at the Astrophysics and Astrochemistry Laboratory at NASA’s Ames Research Center in Mountain View, Calif., a team of researchers have managed to simulate the extreme conditions of interstellar space in order to study the longevity of complex organic compounds. The researchers exposed an icy sample of pyrimidine (an aromatic organic compound that contains four carbon and hydrogen atoms and two nitrogen) to cryogenic temperatures just 10 degrees above absolute zero and irradiated it with high-energy ultraviolet photons, simulating the radiation environment of interstellar space. “Molecules like pyrimidine have nitrogen atoms in their ring structures, which makes them somewhat wimpy,” says Scott Sandford, a senior member at the Astrophysics and Astrochemistry Laboratory at Ames. “As a less stable molecule, it is more susceptible to destruction by radiation, compared to its counterparts that don’t have nitrogen. We wanted to test whether pyrimidine can survive in space, and whether it can undergo reactions that turn it into more complicated organic species, such as the nucleobases uracil, cytosine, and thymine.”

The researchers noticed that instead of being dissolved completely from the intense radiation, the pyrimidine broke down to simpler compounds which then recombined to form uracil, cytosine, and thymine, which are some of the nucleobases found in the DNA and RNA molecules. The reason for these results, according to the researchers, was due to the cryogenic conditions that the pyrimidine was exposed in the first place for the duration of the experiment. While frozen in ice, pyrimidine was much more resilient to the destructive effects of ultraviolet radiation than what it would be if it were in a gas phase, similar to what is seen in open space. This mechanism is not unlike what is observed in dense interstellar clouds, where organic compounds tend to stick onto the icy microscopic grains of interstellar dust, thus protecting themselves from cosmic and stellar irradiation. “We have demonstrated for the first time that we can make uracil, cytosine, and thymine, all three components of RNA and DNA, non-biologically in a laboratory under conditions found in space,” comments Michel Nuevo, member of the Ames Astrophysics and Astrochemistry Laboratory. “We are showing that these laboratory processes, which simulate conditions in outer space, can make several fundamental building blocks used by living organisms on Earth.”
As the advent of exoplanetary research during the last two decades has shown, the Universe is replete with planets, some of which could turn out to be similar to our own. But even more importantly, the results of the simulations by the NASA Ames team, as well as the recent observations from ALMA, provide valuable insights about the prevalence of the necessary materials for life throughout the Cosmos. “From the study of exoplanets, we know the Solar System isn’t unique in its number of planets or abundance of water,” comments Öberg. “Now we know we’re not unique in organic chemistry. Once more, we have learnt that we’re not special. From a life in the Universe point of view, this is great news.”
Indeed, from a cosmic perspective it may be that in the end—to paraphrase the late Carl Sagan—life is just a way for the Universe to know itself.
Want to keep up-to-date with all things space? Be sure to “Like” AmericaSpace on Facebook and follow us on Twitter: @AmericaSpace




Yet another EXCELLENT article Leonidas! As is so often the case, the information is at once very exciting, and very disappointing. Very exciting when one considers the incredible possibilities of life in our solar system, and far beyond. Very disappointing when one considers the appalling apathy of the overwhelming majority of individuals towards anything that does not involve sports, “reality” TV, electronic toys and trinkets, etc. It is extremely frustrating to many who know that we are on the cusp of discoveries that will change our entire world view and perception of ourselves, yet we would rather fixate on the latest antics of some pop culture superstar. Also, thank you very much Leonidas for reminding me of the mystic voodoo of “the Black Arts” – organic chemistry. Yikes! Give me some good ‘ol thermodynamics or fluid flow mechanics any day! 🙂
Hello Karol, it’s great hearing from you again!
Indeed, save for the actual discovery of alien life, I cannot think of anything more exciting than the continuous discoveries of ever smaller potentially habitable exoplanets and the detection of the building blocks of life in the Universe! And of course, I share your frustration about the public’s general apathy towards space exploration – I have written about it in the past here on AmericaSpace and may do so in the future as well. It’s funny, but I just had this discussion earlier today. I went with my girlfriend to the Planetarium here in Athens, to watch the screening of a show that among other things, dealt with the great practical value of space exploration to our everyday lives and we were later both wondering as to why the public is unaware of these things. My girfriend asked as to why, out of all places, people in the US where all the space action is, can be so ignorant about the benefits of space. I didn’t have an answer to give…
As for your agony over the intricacies of organic chemistry, rest assured that I had a similar one concerning math and physics! I still get these nightmares from time to time, where I find myself locked in high school trying to solve some math exams that I don’t know anything about…:)
Once life is detected (and relatively soon), I fear the reaction among the general population may be, “Oh, yeah” and continue to engage in the mundane. It will only be those among us who wonder in awe and contemplate the possibilities.
Chelyabinsk indicates you are right. Humans are mostly stupid and we may be too stupid to survive. Biological life might always self-destruct when it evolves technology. Seems like the most probable answer to the question of why it is so quiet out there.
That’s one of the possible answers to the question of ‘are we alone?’ As the late Albert Einstein so succinctly remarked at one point: ‘Two things are Infinite: the Universe and human stupidity; and I’m not so sure about the first!’
Still, as a species we also have the capacity to act smart and wise in equal measure, if we choose to. I’d like to think that this quality could be the deciding factor for our eventual survival and prosperity in the long run.