
But the Moon that pulls the tides, and the Moon that controls the menstrual periods of women, and the Moon that touches the lunatics, she is not the mere dead lump of the astronomist. . . . When we describe the Moon as dead, we are describing the deadness in ourselves. When we find space so hideously void, we are describing our own unbearable emptiness.
— D.H. Lawrence, “Phoenix: The Posthumous Papers of D.H. Lawrence,” pt. 4, 1930
One of the themes permeating many of English writer David Herbert Lawrence’s works is the stark criticism of prevailing conventional and conservative views of his time. Yet even the acclaimed artist could not have possibly known that his introspective contemplations of our natural satellite as a dynamic world would turn out to be closer to reality than previously thought.
Being an airless celestial body, the Moon was long thought to be drier than the driest places on Earth. Analysis of the lunar rock samples brought back by the astronauts of the Apollo missions during the 1960s and ’70s further strengthened that belief, by revealing scant traces of hydrated minerals in the lunar regolith, which were quickly thought to be the result of contamination from Earth. Similar studies of the samples brought back by the unmanned Soviet Luna 24 in 1976, which showed them to contain about 0.1 percent water by mass, were largely ignored as well. It took a series of very detailed measurements from a new wave of robotic lunar exploration missions during the last 20 years for these long-held views of the Moon as a completely waterless body to finally be overturned. Two of these missions in particular, the Indian Space Research Organization’s Chandrayaan-1 spacecraft and NASA’s Lunar CRater Observation and Sensing Satellite, or LCROSS, being launched in 2008 and 2009 respectively, finally confirmed the existence of huge amounts of water ice deposits at the bottoms of over 40 of permanently shadowed craters near the Moon’s polar regions.
In the absence of an atmosphere, any water molecules on the surface of the Moon would be quickly dissolved by sunlight into its constituent elements. Yet astronomers had theorised since the early 1960s that water ice could exist in permanently shadowed craters near the Moon’s polar regions. Since the Moon’s axis of rotation is almost perpendicular to the plane of the ecliptic, showcasing a tilt of only 1.5 degrees, astronomers had hypothesized that during the Moon’s rotation any deep craters that would be in proximity to the lunar poles would receive no sunlight at all for millions or even billions of years. Consequently, the temperatures inside these regions could steadily remain as low as -454°F (-270 °C), essentially making them cold traps for any volatile elements that would be deposited there.
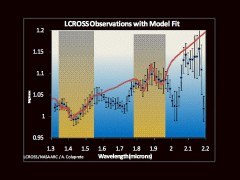
The LCROSS and Chandrayaan-1 missions turned these speculations into fact, by clearly detecting the presence of water and hydroxyl in abundant quantities near the lunar polar regions. “For silicate bodies [like the Moon], such features are typically attributed to water and hydroxyl-bearing materials,” says Dr. Carle Pieters, a planetary scientist from Brown University at Providence, R.I., and Principal Investigator of NASA’s Moon Mineralogy Mapper imaging spectrometer onboard Chandrayaan-1, which detected the water and hydroxyl absorption lines at infrared wavelengths. “When we say ‘water on the Moon,’ we are not talking about lakes, oceans or even puddles. Water on the Moon means molecules of water and hydroxyl that interact with molecules of rock and dust specifically in the top millimeters of the Moon’s surface.” Although these findings wouldn’t qualify our nearest celestial neighbor as a “water world” like Earth, they nevertheless showed that it was hardly a boring and useless chunk of rock, as previously thought. “The emerging picture from many experiments on several different lunar missions indicates that the creation, migration, deposition and retention of vast amounts of water are occurring on the Moon,” writes Dr. Paul Spudis, a Senior Staff scientist at the Lunar and Planetary Institute in Houston, Texas. “Such an astounding result was totally unexpected by most lunar scientists, including myself … The quantity of water present at the lunar poles is significant; at the north pole alone, the 600 million metric tons of water there – turned into rocket fuel – is enough to launch the equivalent of one Space Shuttle (735 mT of propellant) per day for over 2000 years.”

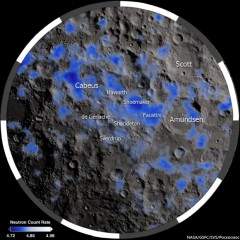
More interestingly, besides the mere presence of these water ice deposits, LCROSS revealed the presence of several organic compounds as well, which were mixed with the ice. Part of the robotic spacecraft’s mission was the LCROSS Impact Cratering Experiment, which involved the crashing of its Centaur upper rocket stage into the Cabeus crater, located approximately 100 km from the lunar south pole, with the goal of analysing the spectra of any sunlit material that would be ejected high above the permanently shadowed crater floor. The analysis of the ejected material’s spectra showed some unexpected traces of other volatile elements as well, besides hydroxyl. “The results from LCROSS were surprising in several ways, not just due to water on the Moon but [because of] other substances: 5.7% carbon monoxide, 1.4% molecular hydrogen, 1.6% calcium, 1.2% mercury, 0.4% magnesium,” writes Dr. Arlin Crotts, a professor at the Columbia University’s Department of Astronomy in New York. “Sulfur is detected as hydrogen sulfide and sulfur dioxide, at levels 1/6th and 1/30th of water, respectively. Nitrogen is seen within ammonia, at 1/16th water’s abundance. Trace amounts (less than 1/30th of water) are detected for ethane, carbon dioxide, methanol, methane and hydroxyl. Volatiles compose at least 1/8th of the soil mass. The poles differ radically from any part of the Moon we have visited or sampled.”
The mechanism by which water ice and organic compounds could be delivered to the Moon’s poles was long thought to be through cometary impacts, considering that many of the substances detected on the Moon’s Cabeus crater by LCROSS are also known to be the main ingredients of comets. According to most scientists, similar cometary impacts on the primordial Earth may have also played a key role in seeding our planet with the necessary building blocks of life. Besides cometary impacts, the solar wind, which is composed of ionised hydrogen, is also suspected to be a source for the volatile elements found on the lunar surface, through its interaction with the oxygen-rich lunar regolith. Yet, according to a recently published study in the scientific journal Icarus, the organics found on the lunar poles could have also been produced in-situ, through the Moon’s constant irradiation by galactic cosmic rays.
http://www.youtube.com/watch?v=Baf3iX58y4EVideo Credit: NASA/ Goddard Space Flight Center
Cosmic rays are very high energy radiation that permeates all of space, coming from all directions from outside of the Solar System. They are mainly consisted of high-energy protons and are thought to originate inside supernova explosions and other high-energy phenomena in the Universe. Although best known for their harmful effects to life, cosmic rays can also act as an energy source for the synthesis of organic compounds from inorganic matter. “Laboratory experiments have repeatedly shown that both hard ultraviolet and energetic protons can stimulate organic synthesis,” writes Dr. Paul G. Lucey, a faculty member at the University of Hawaii’s Institute for Geophysics and Planetology and co-author of the study. “Ultraviolet irradiation of simple mixtures of carbon, hydrogen, oxygen and nitrogen-bearing ices such as water and carbon dioxide (even without nitrogen-bearing ice), produces simple organic molecules. Similar experiments using high energy protons, analogous to galactic cosmic ray protons, also result in the production of simple organics. Both these energy sources are available at the lunar poles. The lunar surface, even the polar surface, is illuminated by scattered interstellar Lyman a UV radiation, and also by galactic cosmic ray protons.”
The study’s team, led by Sarah Crites, a graduate student at the University of Hawaii at Manoa, ran a series of Monte Carlo simulations to estimate the flux of galactic cosmic rays at the Moon’s surface and their level of penetration inside the lunar regolith, based on these previous laboratory experiments. In order to check the validity of their models, they compared their results with the radiation dose measurements that were taken at the vicinity of the Moon with the Cosmic Ray Telescope for the Effects of Radiation, or CRaTER instrument, onboard NASA’s Lunar Reconnaissance Orbiter spacecraft. Although not a perfect match, the team’s results were found to be in good agreement with those from LRO. “Our surface dose rate is lower than the CraTER results, and fall slightly outside the standard deviation from the average from CraTER,” writes the team in their study. “These differences may stem from our simple scaling to include heavy ions [in the simulation, besides protons], which does not necessarily account for all the physical processes related to heavy ion interactions in the lunar soil. However, our results are within 25 percent of the CraTER results, and suggest that [the Monte Carlo simulations] can be applied to studies of the general radiation environment at the Moon over lunar [geologic] history, when the modest difference between measurement and model is taken into account.”
The team’s results suggest that a small amount of volatiles on the Moon’s polar regions, irradiated with galactic cosmic rays over a period of approximately a billion years, could have produced the organic compounds that were detected with LCROSS’ instruments at the bottoms of the Moon’s polar craters. “By comparing our particle flux simulations with experimental data, we find that proton flux from galactic cosmic rays provides ample exposure to plausibly drive organic synthesis over timescales close to, or probably much less than the age of the surface and the longevity of the cold traps,” concluded the team during a presentation of their study. “In addition, since many organics are more refractory than ices, the organics produced may remain even as the extent of cold traps changes with time.”
The in-situ production of organic molecules on the Moon’s surface, if proven, will open up a whole new and exciting area of study into one of science’s most fundamental questions: the origin of life itself. “The generation of complex organic molecules is an important topic of research for the origin of life,” comments Dr. Spudis on Crites’ team’s results. “As we continue to study the Moon, we find that it offers much more than one might suspect at first glance … The poles of the Moon are thus a natural laboratory for the study of one of the early processes in Solar System history – the creation of complex organic substances from the more primitive and simple elements and compounds. In this sense, the pre-biotic organic chemistry of the lifeless and barren Moon serves the cause of the study of life’s processes and origin…Far from being a mere echo of some previous space glory, a return to the Moon to undertake new scientific studies, new exploration and to develop a wholly new set of technologies impacts all of space science and exploration in many different and unexpected ways.”
Video Credit: NASA/Goddard Space Flight Center
Sadly, the White House’s current space policy precludes the lunar surface as a future destination for U.S. astronauts, viewing the Moon as a “been there, done that” object instead. Having successfully conducted six manned lunar landings during the Apollo program, NASA also currently sees any further human lunar surface exploration as being wasteful and unnecessary on the agency’s road to Mars. “The samples returned from the lunar surface by Apollo were devoid of organic material,” comments Dr. Lucey. “However, the equatorial regions of the Moon sampled by Apollo and [the Soviet] Luna are not representative of the Moon as a whole. The lunar poles harbor a microenvironment which possess conditions utterly unlike those of the lunar equator. These conditions may allow in situ production of organics on the Moon from indigenous inorganic material. If this is the case, the Moon may allow field-testing of models of inorganic synthesis which have been invoked for many surfaces in the Solar System, and even interstellar clouds.”
Indeed, seen in this light, a human return to the Moon to build on the legacy of the Apollo missions of the 20th century may turn out to be one of the best investments that we could make as a space-faring civilisation during the first quarter of the 21st.
Want to keep up-to-date with all things space? Be sure to “Like” AmericaSpace on Facebook and follow us on Twitter: @AmericaSpace




Leonidas’s concluding statement underscores the need to explore the Moon. We are foolish and extremely short-sighted not to continue lunar exploration.