
How did life on Earth begin? The question of the origin of life on our planet is as old as the human species itself. The lack of a definitive answer to this fundamental mystery has given rise to a host of different hypotheses which aim to explain the appearance of the first self-replicating molecules on the primordial Earth, which formed the basis for all the biological diversity we see today. New research now comes to add more pieces to the puzzle by showing that some of the building blocks of DNA, which is the molecule of all terrestrial life, could have been previously cooked up in interstellar space inside the circumstellar envelopes of dying carbon-rich stars, before being incorporated eventually into the newly formed Earth.
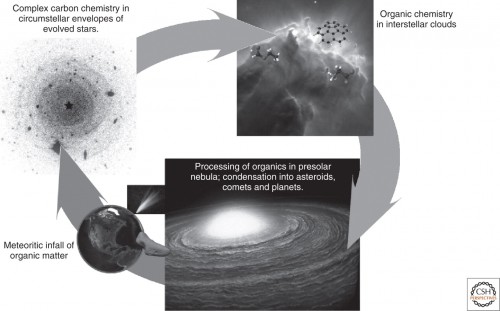
Life as we know it arose from the intricate chemical reactions between organic molecules – long chains of carbon atoms which bonded together with oxygen, hydrogen, nitrogen, sulfur, and phosphorus atoms that eventually formed macromolecules like carbohydrates, lipids, amino acids, proteins, and nucleic acids – the building blocks of life itself. But what were the exact steps in the transition between the simpler pre-biotic ingredients and the first self-replicating molecules of DNA and RNA in the primordial Earth? One of the leading long-standing hypotheses which have been strengthened by the famous Miller–Urey experiment, posits that these evolutionary first steps towards life took place within the primordial waters of the early Earth where a combination of the right environmental conditions and chemical reactions ultimately led to the emergence of life. Yet, many scientists have argued that when our planet was formed approximately 4.5 billion years ago, it may had an atmospheric composition that was different from what was assumed in the Miller–Urey experiment. In addition, surface conditions could have been too hot and volatile due to the constant bombardment from comets and asteroids, which was very common in the early history of the Solar System, to have allowed for the development of a ‘primordial soup’ and a subsequent in-situ emergence of life.
A second school of thought has argued that the necessary buildings blocks for life weren’t developed in the early Earth itself, but were cooked up in space instead only to be delivered later to our planet through asteroid and cometary impacts. During the early 20th century, the formation of complex organic compounds in space were thought to be highly unlikely. Contrary to these beliefs, astronomers subsequently discovered that organic molecules were indeed everywhere in space – from the surfaces and atmospheres of planets and moons in our Solar System, to the rarefied gas and dust of the interstellar medium. The advent of infrared astronomy during the last 50 years has revealed that the latter is indeed rich in organic chemistry, featuring an ever-growing list of hundreds of organic molecules many of which were found to be surprisingly complex, like vinyl alcohol, octatetraynyl anions, anthracene molecules and gycline just to name a few. In recent years, scientists have also been discovering large quantities of organic material in places that were previously thought to be too hostile, like the vicinity of the supermassive black hole Sagittarius A* at the center of the Milky Way and the circumstellar disks of newly formed stars, as detailed in a previous AmericaSpace article. Furthermore, a type of meteorites, called carbonaceous chondrites, that have been discovered on many meteorite impact sites across the globe, were found not only to be rich in organic compounds but some of them, like the famous Murchison meteorite, were also found to carry amino acids as well as nucleobases like adenine and guanine that were of extraterrestrial origin. Adenine and guanine in particular, along with cytosine, thymine and uracil, are the five primary building blocks that combine in pairs to form the helical structures of DNA and RNA – the very molecules of life itself.

One question that naturally arises from these findings, is where and how exactly these essential building blocks of life are created in space? A new study that was led by a team of researchers at the Lawrence Berkeley National Laboratory in California and the University of Hawaii at Manoa, aims to shed more light on this question, by showing that the chemical elements that make up the nucleobases of the DNA and RNA molecules can be created in the hot environments of the circumstellar envelopes around carbon-rich stars that have reached the end of their lives. More specifically, the researchers focused on a subset of nitrogen-containing, carbon-rich organic compounds, called Polycyclic Aromatic Hydrocarbons, or PAHs for short. The latter have been found in great abundances in a gas phase throughout the interstellar medium, as well as in comets and asteroids. On Earth PAHs are the common byproducts of the burning of fossil fuels like coal and petroleum. From an astrobiological perspective they are very important, since they are considered to have been key ingredients of the primordial soup from which life arose on early Earth. With that in mind, the researchers utilised the Advanced Light Source, a synchrotron particle accelerator which is located at the Lawrence Berkeley National Laboratory, to simulate the high temperature conditions that are found in the circumstellar envelopes of carbon-rich stars in order to study the chemical pathways leading from simpler organic compounds to polycyclic aromatic hydrocarbons and other complex organics in these environments, which haven’t been probed before. To that end, the research team led by Dorian Parker from the University of Hawaii at Manoa and postdoctoral fellow at the NASA Astrobiology Institute in California, used laser pyrolysis to heat a sample of gaseous meta-pyridyl radicals inside a silicon carbide nozzle at temperatures of 780 Kelvin, in the presence of acetylene (another astrobiologically important and cosmically abundant molecule) which acted as a catalyst. The results of the experiment showed that through gas-phase reactions, the meta-pyridyl radicals when combined with acetylene, produced quinoline and isoquinoline – two species of polycyclic aromatic nitrogen heterocycles, or PANHs for short, which in essence are polycyclic aromatic hydrocarbons whose carbon atoms have been replaced by nitrogen.
Even though previous laboratory experiments have shown that polycyclic aromatic hydrocarbons can potentially undergo similar reactions towards more complex organics like amino acids and even nucleobases after being stuck to icy grains in the cold interstellar medium, the new study by Parker’s team was the first to simulate the hot conditions of stellar envelopes. “This is the first time anyone’s looked at a hot reaction like this,” says Musahid Ahmed, a scientist at the Lawrence Berkeley National Laboratory and member of the research team. “It’s not easy for carbon atoms to form rings that contain nitrogen. But this new work demonstrates the possibility of a hot gas phase reaction, a ‘cosmic barbecue’.” Furthermore, the specific chemical reactions that were studied in the team’s research, are only possible at high-temperature environments like those found in the vicinity of dying carbon-rich stars. “There’s an energy barrier for this reaction to take place, and you can exceed that barrier near a star or in our experimental setup,” says Ahmed. “This suggests that we can start looking for these molecules around stars now.”

One key characteristic of PANHs compared to the comparatively simpler PAHs, is that the former can interact more strongly with water ice dust grains in the interstellar medium, which could better shield them from the destroying effects of ultraviolet stellar irradiation. This way, polycyclic aromatic nitrogen heterocycles can more readily condense into cold dust grains and be later reprocessed into more complex molecules like nucleobases. “In summary, our combined experimental and computational study provides compelling evidence and manifests a hitherto overlooked gas-phase synthetic route leading to the formation of a key class of bio-relevant molecules in the inner regions of circumstellar envelopes of carbon stars – nitrogen-containing PAHs (NPAHs) with quinoline and isoquinoline being their prototypical representatives,” conclude the researchers in their study, which was recently published at The Astrophysical Journal. “Once synthesized in the circumstellar envelopes and eventually incorporated into interstellar grains in cold molecular clouds, NPAHs can be functionalized leading to nucleobase-type structures like purines, as sampled in the Murchison meteorite. This scenario provides a strong alternative to the terrestrial in-situ formation of NPAHs and their functionalized counterparts such as nucleobases on early Earth, which have been proposed to involve solutions of hydrogen cyanide and ammonia condensation-dehydration sequences encompassing formamide and possibly spark discharge of methane and nitrogen in the presence of water ice.”
While the results of the study are only suggestive, they nevertheless provide a fascinating new piece of the puzzle in the quest to understand the intricate pathways that eventually led from chemical to biological evolution here on Earth and possibly elsewhere in the Universe as well. “We don’t know if this [organic] material [on meteorites] really was important for the origin of life,” had commented in a past interview for the Astrobiology Magazine, Dr. Pascale Ehrenfreund, now a research professor at the George Washington University’s Space Policy Institute, in Washington, DC. “But since we know that it is extraterrestrial and it arrived intact on the early Earth, we have a sample of material that could have been important to further processing and for the build up of complexity”.
The search for how life started here on Earth is as painstaking as the search for life elsewhere in the Universe. Could it be that extraterrestrial life, if and when it is ever discovered, could be sharing the same essential building blocks with its terrestrial counterpart, in a vast Cosmos that is itself rich in the stuff of life?
Want to keep up-to-date with all things space? Be sure to “Like” AmericaSpace on Facebook and follow us on Twitter: @AmericaSpace




Καλημερα Λεωνιδα ελπιζω να εισαι καλα,το αρθρο τελειο,φιλια!!!!!!!!!!!!!!!!
The building blocks for DNA absolutely are found in space and also on the periodic table. This doesn’t come anywhere near resolving the issue of life. Nobody can sincerely study the mechanism of life without being in awe.
I’m waiting for this headline…
“Random memory results in intelligent program”
I hope Galacticus is not in our part of the galaxy looking for a meal. And then there is the “Solaris” concept of planet beings. The Star Trek episode about the giant life sucking Amoeba. And even the movie Fantastic Voyage, which instead of a human body might well have taken place inside a giant organism on some ocean moon.